
These highly specialized and asymmetric cells form elaborate axonal and dendritic arbors, with some axons extending great distances (e.g., axons in a blue whale can reach a length of 30 meters Smith, 2009). Neurons present a dramatic example of cell polarization. These experiments reveal a new-found membrane trafficking pathway, for SV proteins, in classically polarized mammalian neurons and provide a glimpse at the first steps of SV biogenesis. 
Moreover, we observed that SV constituents were first delivered to the presynaptic plasma membrane before incorporation into SVs. However, even moderate overexpression resulted in the spillover of SV proteins into dendrites, potentially explaining the origin of previous non-polarized transport models, revealing the limited, saturable nature of the direct axonal trafficking pathway. In sharp contrast to the selective retention model, both proteins selectively and specifically entered axons with minimal entry into dendrites. For these studies, the SV reporter constructs were expressed at carefully controlled, very low levels. Here, we used the RUSH (retention using selective hooks) system, in conjunction with HaloTag labeling approaches, to study the egress of two distinct transmembrane SV proteins, synaptotagmin 1 and synaptobrevin 2, from the soma of mature cultured rat and mouse neurons. The leading model posits that these proteins are randomly trafficked throughout neurons and are selectively retained in presynaptic boutons. Yet, how SV proteins are sorted to presynaptic nerve terminals remains the subject of debate. There are only differences between fast secreting neurotransmitters (Acth, dopamine) and slow releasing neuropeptides from neuroendocrine cells.Neurotransmitter-filled synaptic vesicles (SVs) mediate synaptic transmission and are a hallmark specialization in neuronal axons. The specific capacitance is mainly determined by the thickness and dielectric constant of the phospholipid bilayer membrane and is similar for intracellular organelles and the plasma membrane. This can be measured by increases in membrane capacitance (Cm). Another direct measure of exocytosis is the increase in membrane area due to the incorporation of the secretory granule or vesicle membrane into the plasma membrane. Meaning that upon stimulation, many neurotransmitters are being released into the synaptic cleft.īut there a thing called membrane capacitance. 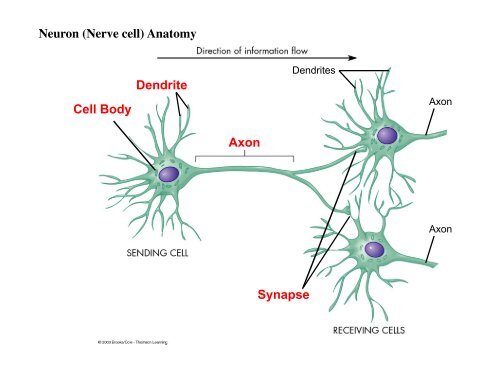
In the process, they cause changes in the permeability of the cell membrane to specific ions, opening up special gates or channels which let in a flood of charged particles (ions of calcium, sodium, potassium, and chloride). These chemicals then bind to chemical receptors in the dendrites of the receiving (post-synaptic) neuron. 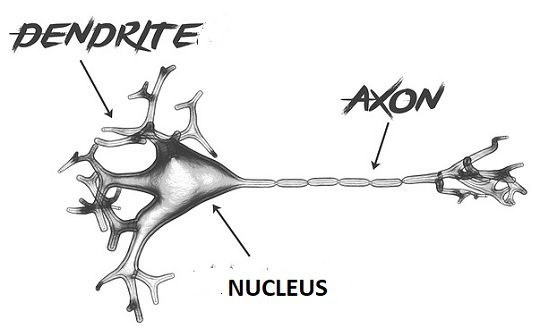
When stimulated by an electrical pulse, neurotransmitters of various types are released, and they cross the cell membrane into the synaptic gap between neurons. The more signals sent between two neurons, the stronger the connection grows. The connections between neurons are not static, though, they change over time. Meaning that a certain neuron sends multiple kinds of signals by sending different kinds of neurotransmitters.įunctionally related neurons connect to form neural networks (also known as neural nets or assemblies). Each neuron may be connected to up to 10,000 other neurons, passing signals to each other via as many as 1,000 trillion synaptic connections.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
